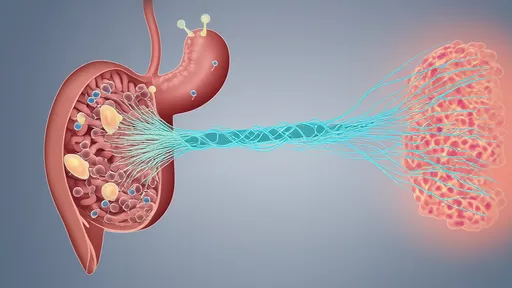
Recent advances in neuroimmunology have unveiled a sophisticated bidirectional communication network between the gut and the brain, mediated largely by the vagus nerve. This tenth cranial nerve, often described as the body's superhighway for gut-brain signaling, is now recognized as a critical modulator of inflammatory responses. Emerging research suggests that the vagus nerve doesn't merely transmit signals but actively participates in decoding and regulating gut-derived inflammation through what scientists term "closed-loop control" - a continuous feedback system that maintains immunological balance.
The gut-brain axis has long fascinated researchers, but only in the past decade have we begun to understand the vagus nerve's precise role in inflammation regulation. Unlike traditional views of neural signaling as one-directional commands, new evidence reveals a dynamic conversation where gut microbes, immune cells, and neural pathways engage in constant dialogue. This discovery fundamentally changes our understanding of how chronic inflammatory conditions develop and how they might be treated.
Molecular interpreters along the neural pathway
Specialized receptors along the vagus nerve act as molecular interpreters, translating inflammatory signals from the gut into neural language. These receptors detect cytokines, bacterial fragments, and other immune molecules, converting them into electrical impulses that travel to the brainstem. What's remarkable is the system's precision - different patterns of neural firing correspond to specific types and intensities of inflammation, allowing for targeted responses.
The brain doesn't merely receive these signals passively. Nucleus tractus solitarius, the primary termination site of vagal afferents, functions as a sophisticated processing center that integrates inflammatory information with other bodily states. From here, signals diverge to multiple brain regions including the hypothalamus and amygdala, linking gut inflammation to metabolic regulation, stress responses, and even emotional states.
The efferent arm completes the loop
Completing the circuit, vagal efferents carry the brain's responses back to the gut and other organs through what's known as the cholinergic anti-inflammatory pathway. This isn't a simple on-off switch but a nuanced modulation system. Acetylcholine release from vagal fibers binds to specific receptors on immune cells, particularly macrophages, dampening their production of pro-inflammatory cytokines without suppressing protective immunity.
This closed-loop system explains why vagus nerve stimulation shows promise in treating conditions ranging from rheumatoid arthritis to depression. The therapeutic effects aren't merely about increasing or decreasing neural activity but restoring the system's natural capacity for dynamic equilibrium. Researchers are now mapping how different stimulation parameters can mimic the body's own regulatory patterns.
Microbial partners in neural signaling
Gut microbiota play an unexpected role as active participants in this neural dialogue. Certain bacterial strains produce neurotransmitters and metabolites that directly interact with vagal receptors. For instance, Lactobacillus species have been shown to increase vagal firing rates, while pathogenic microbes often trigger distinct inflammatory signaling patterns. This microbial-neural crosstalk suggests that probiotics might exert some of their benefits through vagus-mediated mechanisms.
The vagus nerve's ability to distinguish between microbial friends and foes adds another layer of complexity to gut-brain communication. Early evidence indicates that the nerve doesn't simply respond to the presence of bacteria but interprets the ecological context - whether certain microbial populations are appropriately balanced and properly located within the intestinal environment.
Clinical implications and therapeutic horizons
Understanding this closed-loop regulation opens new avenues for treating inflammatory diseases. Bioelectronic medicine approaches aim to develop "smart" vagus nerve stimulators that can detect inflammatory patterns and deliver precisely timed counter-signals. Unlike drugs that bathe the body in molecules, these devices would work with the body's natural control systems.
Nutritional interventions also take on new significance in light of vagus nerve decoding. Certain dietary components appear to enhance the gut-brain axis's regulatory capacity, not just by altering microbiome composition but by optimizing the neural signaling environment. Omega-3 fatty acids, polyphenols, and fermentable fibers all show potential for strengthening vagus-mediated inflammation control.
The discovery of this sophisticated decoding system challenges traditional distinctions between neurology, immunology, and gastroenterology. As research progresses, we're learning that inflammation isn't merely something the body suffers but a language it uses - with the vagus nerve serving as both interpreter and mediator in an ongoing conversation between gut and brain.

By /Jul 28, 2025
By /Aug 5, 2025

By /Jul 28, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025
By /Jul 28, 2025
By /Jul 28, 2025