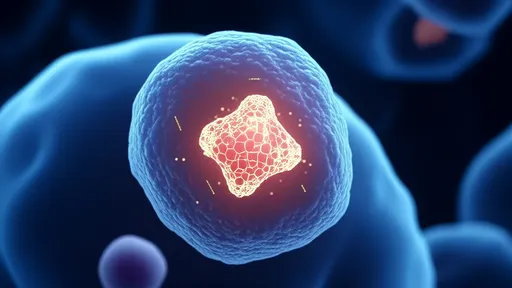
In a groundbreaking development for cellular biology and medical diagnostics, researchers have successfully employed nanodiamond quantum sensors to measure localized temperatures within living cells with unprecedented precision. This innovative approach, dubbed "diamond quantum thermometry," leverages the unique quantum properties of nitrogen-vacancy (NV) centers in diamond crystals to detect subtle thermal fluctuations at the subcellular level. The technique promises to revolutionize our understanding of metabolic processes, disease mechanisms, and therapeutic interventions at scales previously inaccessible to conventional thermometry.
The quantum advantage in cellular thermometry stems from diamond's remarkable physical properties. When engineered with specific atomic-scale defects, these carbon crystals become exquisitely sensitive to temperature variations through changes in their electron spin resonance. Unlike traditional temperature probes that often disrupt cellular function or provide averaged measurements across entire cell populations, nanodiamond sensors can be precisely positioned within organelles or cytoplasmic regions while causing minimal interference to biological activity. Early experiments have demonstrated temperature resolution better than 0.1°C with spatial precision under 200 nanometers—a capability that allows researchers to map thermal gradients across individual mitochondria or other subcellular structures.
What makes this technological leap particularly significant is the dynamic nature of cellular temperature regulation. Metabolic activity, ion channel operations, and biochemical reactions all generate or absorb heat in ways that reflect the cell's functional state. Previous limitations in measurement technology forced scientists to treat cells as isothermal systems, potentially missing critical thermal signatures of disease states or drug responses. The diamond quantum thermometry approach reveals that temperature variations within single cells can exceed 1°C during normal activity—a finding that overturns longstanding assumptions about thermal homogeneity in cellular environments.
Implementation of this technique involves embedding fluorescent nanodiamonds (typically 50-100nm in size) into target cells through either endocytosis or microinjection. These diamond particles contain NV centers whose spin states can be optically initialized and read out using laser pulses, while microwave frequencies probe the temperature-dependent shifts in their energy levels. The entire measurement process occurs without physical contact or electrical interference, maintaining the cell's viability for extended observation periods. This non-invasive characteristic represents a major advancement over earlier methods that required inserting metallic probes or fluorescent polymers into cells.
Applications in cancer research have already yielded compelling results. Tumor cells typically exhibit elevated metabolic rates compared to healthy tissue, but the new quantum sensing technique has revealed unexpected spatial patterns in this thermal activity. Researchers observed distinct "hot spots" near the nuclei of certain cancer cell lines that correlate with specific phases of the cell cycle, suggesting temperature could serve as a novel biomarker for proliferation states. Pharmaceutical companies are particularly interested in using this approach to monitor how experimental cancer drugs affect the thermal profiles of both malignant and normal cells during preclinical testing.
The technology's potential extends beyond basic research into clinical diagnostics. Early detection of infections or inflammatory conditions might become possible by tracking characteristic changes in immune cell temperatures during their activation. Neurologists speculate about applying nanodiamond thermometry to study the thermal signatures of neuronal activity, which could provide new insights into seizure mechanisms or neurodegenerative diseases. Some research groups are even exploring whether the technique could eventually be adapted for in vivo use, potentially enabling non-invasive temperature monitoring of deep tissues through strategically injected nanodiamond tracers.
Despite these exciting possibilities, challenges remain in translating diamond quantum thermometry from laboratory benches to widespread adoption. Current systems require sophisticated optical setups with confocal microscopes and microwave generators, limiting accessibility for many research institutions. There are also ongoing investigations into the long-term biocompatibility of nanodiamonds, although preliminary studies suggest they exhibit excellent tolerance in various cell types. Researchers are working to simplify the readout technology and develop standardized protocols that would allow more laboratories to implement this cutting-edge measurement capability.
The convergence of quantum physics and cell biology represented by this advancement highlights how cross-disciplinary approaches can solve previously intractable problems in life sciences. As the technology matures, it may establish intracellular temperature mapping as a standard tool in both research and clinical settings—much like fluorescence microscopy revolutionized cellular imaging decades ago. With several research groups worldwide now refining different aspects of diamond quantum thermometry, the coming years will likely see significant improvements in sensitivity, temporal resolution, and practical usability of these remarkable nanoscale temperature probes.

By /Jul 28, 2025
By /Aug 5, 2025

By /Jul 28, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025

By /Jul 28, 2025

By /Aug 5, 2025

By /Aug 5, 2025
By /Jul 28, 2025
By /Jul 28, 2025